Asimov launches LV Edge Packaging System to optimize lentivirus production
New off-the-shelf system reduces the cost and risk of lentiviral production for cell and gene therapy developers
31 Jan 2024
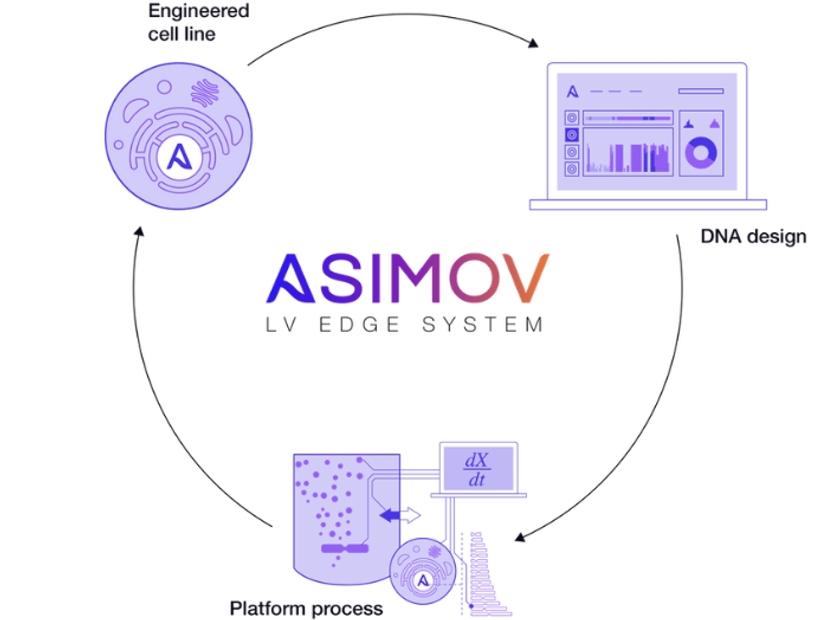
Asimov, a synthetic biology company advancing the design and manufacture of therapeutics, has announced the launch of its LV Edge Packaging System. The ready-to-transfer system minimizes GMP plasmid cost, process complexity, and supply chain risk by stably integrating viral genes into the host cell. This enables lentiviral production from a single-plasmid transfection, in contrast to the standard four-plasmid process.
In lentiviral manufacturing, GMP plasmids account for a substantial proportion of raw material costs. Procurement of these plasmids also introduces additional supply chain risk and process complexity, which can impact both timelines and product variability. By removing the need to transiently transfect three out of the four GMP plasmids, the LV Edge Packaging System reduces manufacturing cost and supply chain risk without compromising speed to market.
The LV Edge Packaging System achieves high harvest titers of over 1E8 TU/mL across multiple chimeric antigen receptor (CAR) transgenes and enables tuneable transgene expression in the transduced cell. The system consists of: (1) A clonal, suspension-adapted packaging cell line with stably-integrated lentiviral genes under inducible control, (2) Model-guided DNA design to optimize transgene expression – powered by Kernel, Asimov's computer-aided design (CAD) software, and (3) Robust, ready-to-transfer processes and protocols across multiple scales.
