Molecular Diagnostics News Update
The very latest molecular diagnostic news, interviews and videos
10 Dec 2015

Molecular diagnostic advances offer the prospect of personalized healthcare
The field of molecular diagnostics, the use of a patients genetic code and molecular techniques, to diagnose disease and illness, is a fascinating one. This round-up of the latest molecular diagnostic news includes advances in clinical next-generation sequencing, infectious disease diagnostics and point-of-care testing.
Thermo Fisher Scientific has entered into a long-term agreement with Novartis and Pfizer to develop and commercialize a multi-marker, universal next-generation sequencing oncology test that will serve as a companion diagnostic for non-small cell lung cancer across multiple drug development programs.
Beckman Coulter Diagnostics has announced the availability of an independent case study, undertaken in Spain, that demonstrates that the company’s DxN VERIS Molecular Diagnostics System* has the potential to improve productivity and turnaround times.
3. QIAGEN Introduces GeneReader NGS System
QIAGEN has announced the start of commercialization activities for its GeneReader NGS System, the first complete Sample to Insight next-generation sequencing (NGS) solution designed for any laboratory to deliver actionable results.
Accurate and prompt diagnoses of Acute Lower Respiratory Tract Infections (LRTIs) is vital to ensure correct treatment plans are in place to improve patient management. Dr Marcus Panning, physician and medical virologist, at The University Medical Centre Freiburg, has selected the Fast-track diagnostics Respiratory pathogens 21 assay to detect LRTIs and effectively diagnose vulnerable patients.
Bio-Rad’s Droplet Digital PCR (ddPCR™) technology identifies viral RNA mutations in clinical samples with greater precision, sensitivity, and reproducibility than quantitative reverse-transcription PCR (RT-qPCR), according to a new study in the Journal of Virological Methods (November 2015).
6. Video: Using OneSeq to Advance the Diagnosis of Genomic Disorders
Professor Orsetta Zuffardi, Ph.D., Department of Molecular Medicine, University of Pavia, describes her laboratory’s work in postnatal and prenatal diagnostics and research into genomic rearrangements. In this video she hopes to use OneSeq technology from Agilent as an alternative to array-CGH in the diagnosis of genomics disorders.
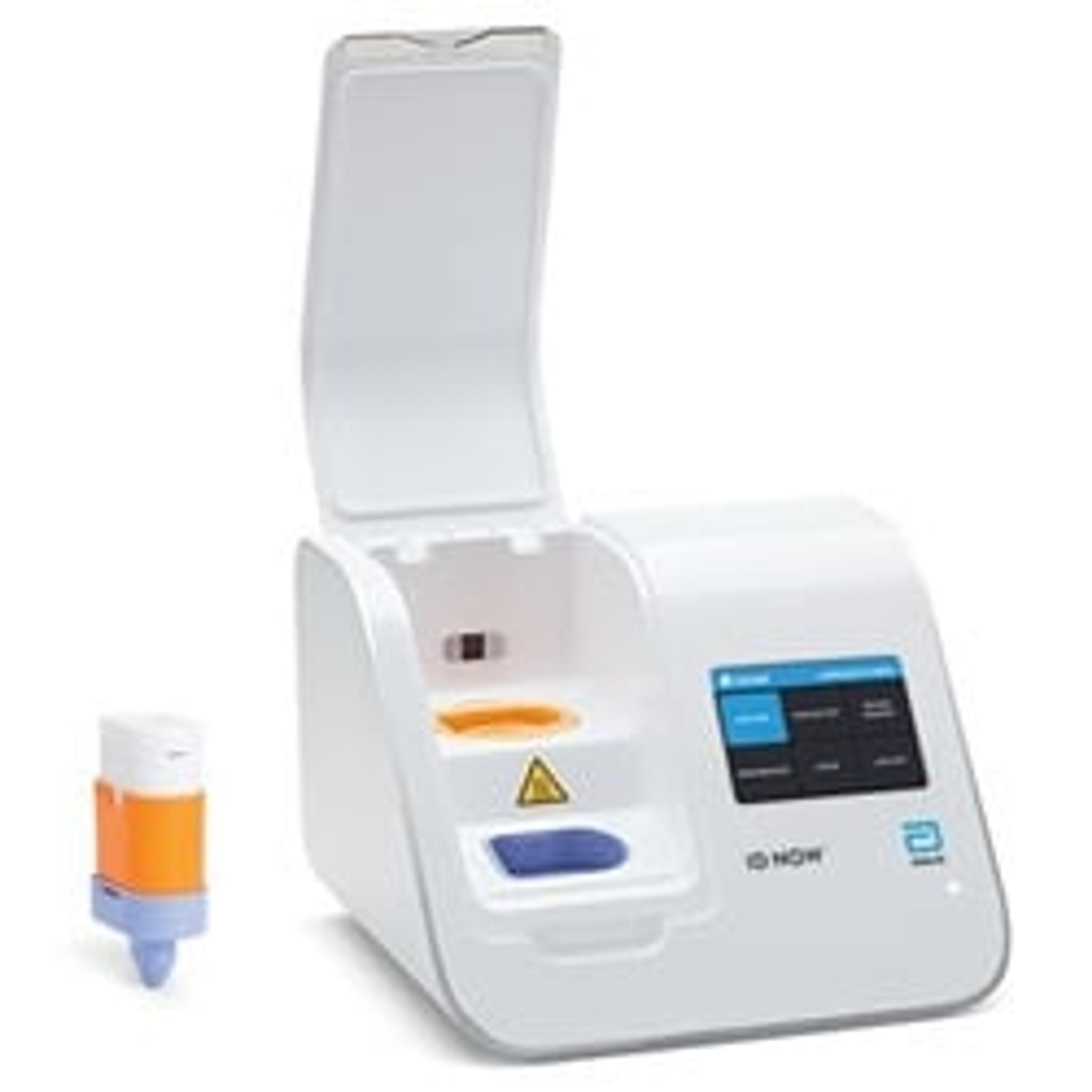
7. Video: Lab Quality Molecular Testing Available at the Point-of-Care: Alere at AACC 2015
The Alere™ i Molecular Platform is not only incredibly easy to use, but also provides high quality, rapid molecular testing in just minutes. Watch this interview with KC McGrath, Respiratory Product Manager, Alere Inc., to find out how this state-of-the-art technology is having such a positive impact on patient care.